Neuro-immune interactions
This facet of my research focuses upon mechanisms through which the brain and immune system communicate with each other. In this respect, protein messengers, called cytokines, released from immune cells inform the brain of infectious/inflammatory events. These cytokines can influence many basic functions of the brain, including neurotransmission, neuroplasticity and the activity of glial cells. In particular, cytokines can activate the brain’s own specialized immune glial cell, the microglia, which ultimately play a fundamental role in regulating the brain’s response to environmental toxins or stressors.
Inflammatory aspects of neurodegeneration with a focus of Parkinson’s
Parkinson’s, Alzheimer’s, multiple sclerosis and other neurodegenerative diseases all have a strong neuroinflammatory component. My present research seeks to determine the role of inflammatory factors, particularly microglia, in Parkinson’s disease. We also have a focus on how environmental toxins might interact with genetic vulnerabilities to cause Parkinson’s.
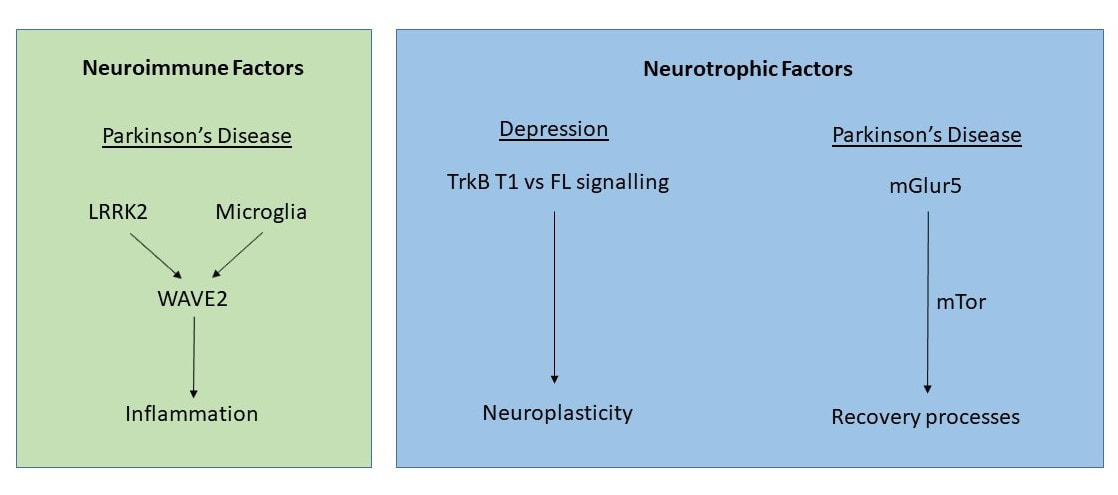
One inflammatory gene in particular, LRRK2, is thought to play an important role in Parkinson’s disease. A current major project in my lab is devoted to understanding how LRRK2 could be involved in controlling microglial interactions with dopamine neurons and specifically, how such interactions can influence neuronal survival or death.
Another major project ongoing in my lab involves the use of a mGlur5 acting drug, CTEP, to promote plasticity of the dopaminergic system in an attempt to cause functional recovery in toxin and genetic based Parkinson’s models. This project has very high clinical relevance.
Stress, neuroplasticity and depression
Many types of stressors exist that range from primarily psychological in nature (psychogenic) to those that impact directly upon physical aspects of individuals (neurogenic) to systemic or immunological stressors, such as cytokines. Some of our current work examines the underlying neurotrophic pathways that are affected by stressors and how targeting these might result in the development of novel antidepressants.
We are particularly interested in how to use immune (e.g. erythropoietin or GM-CSF) or pharmacological agents (e.g. ketamine) to boost neuroplasticity for the treatment of depression. These agents can act by stimulating neurotrophic factors, such as BDNF, that can greatly affect the plasticity of brain circuits that regulate mood. We are currently exploring the differential involvement of BDNF's TrkB.FL and TrkB.T1 receptors in these brain circuits.
This facet of my research focuses upon mechanisms through which the brain and immune system communicate with each other. In this respect, protein messengers, called cytokines, released from immune cells inform the brain of infectious/inflammatory events. These cytokines can influence many basic functions of the brain, including neurotransmission, neuroplasticity and the activity of glial cells. In particular, cytokines can activate the brain’s own specialized immune glial cell, the microglia, which ultimately play a fundamental role in regulating the brain’s response to environmental toxins or stressors.
Inflammatory aspects of neurodegeneration with a focus of Parkinson’s
Parkinson’s, Alzheimer’s, multiple sclerosis and other neurodegenerative diseases all have a strong neuroinflammatory component. My present research seeks to determine the role of inflammatory factors, particularly microglia, in Parkinson’s disease. We also have a focus on how environmental toxins might interact with genetic vulnerabilities to cause Parkinson’s.
One inflammatory gene in particular, LRRK2, is thought to play an important role in Parkinson’s disease. A current major project in my lab is devoted to understanding how LRRK2 could be involved in controlling microglial interactions with dopamine neurons and specifically, how such interactions can influence neuronal survival or death.
Another major project ongoing in my lab involves the use of a mGlur5 acting drug, CTEP, to promote plasticity of the dopaminergic system in an attempt to cause functional recovery in toxin and genetic based Parkinson’s models. This project has very high clinical relevance.
Stress, neuroplasticity and depression
Many types of stressors exist that range from primarily psychological in nature (psychogenic) to those that impact directly upon physical aspects of individuals (neurogenic) to systemic or immunological stressors, such as cytokines. Some of our current work examines the underlying neurotrophic pathways that are affected by stressors and how targeting these might result in the development of novel antidepressants.
We are particularly interested in how to use immune (e.g. erythropoietin or GM-CSF) or pharmacological agents (e.g. ketamine) to boost neuroplasticity for the treatment of depression. These agents can act by stimulating neurotrophic factors, such as BDNF, that can greatly affect the plasticity of brain circuits that regulate mood. We are currently exploring the differential involvement of BDNF's TrkB.FL and TrkB.T1 receptors in these brain circuits.
List of Publications
- Prowse N, Hayley S (2021). Microglia and BDNF at the crossroads of neuropsychiatric illness: Towards a unique trophic phenotype. Neurosci Biobeh Rev, Sep 16;131:135-163.
- Hayley S, Sun H (2021). Neuroimmune multi-hit perspective of coronaviral infection. J Neuroinflam, Oct 13;18(1):231. doi: 10.1186/s12974-021-02282-0.
- Barbara Fenner, Natalie Prowse, Teresa Fortin, Shawn Hayley (2021). A novel dual-hit in vitro model of Parkinson’s disease: WAVE2-dependent progression of microglial inflammatory phenotype. J. Cell Physiol., doi: 10.1002/jcp.30588.
- Zach Dwyer, Melany Chaiquina, Jeffrey Landrigana, Kiara Ayouba, Pragya Shaila, Julianna Rochab, Christie L Childersc, Kenneth B Storey, Dana J. Philpottb, Hongyu Sun a, and Shawn Hayley (2021); The impact of dextran sodium sulphate and probiotic pre-treatment in a murine model of Parkinson’s disease. J. Neuroinfammation Jan 9;18(1):20.
- Zach Dwyer, Chris Rudyk, Kyle Farmer, Sheryl Beauchamp, Pragya Shail, Alexa Derksen, Teresa Fortin, Katelyn Ventura, Carlos Torresd, Kiara Ayoub Shawn Hayley PhD (2021). Characterizing the protracted neurobiological and neuroanatomical effects of paraquat in a murine model of Parkinson’s disease. Neurobiology of Aging, Jan 9;18(1):20
- Hayley S, Hakim AM, Albert PR (2021). Depression, Dementia and Immune Dysregulation. Brain, Dec 5, doi: 10.1093/brain/awaa405.
- Alysia Ross, Viktoria Xing, Ting Ting Wang, Sam Bureau, Teresa Fortin, Hui Zhang, Shawn Hayley, Hongyu Sun (2020). Alleviating toxic α-Synuclein accumulation by membrane depolarization: evidence from an in vitro model of Parkinson’s disease. Mol. Brain, 13(1):108. doi: 10.1186/s13041-020-00648-8.
- Aycheh Al-Chami, Alysia Ross, Shawn P. Hayley, Hongyu Sun. (2020). Early life stress facilities synapse premature unsilencing to enhance AMPA receptor function in the developing hippocampus. J. Neurophysiology, 124(3):815-821.
- Thompson, A.M., Farmer, K., Rowe, E.M., & Hayley, S. (2020). Erythropoietin modulates striatal antioxidant signalling to reduce neurodegeneration in a toxicant model of Parkinson’s disease. Mol Cell Neurosci, 109:103554. doi: 10.1016/j.mcn.2020.103554.
- Zach Dwyer, Chris Rudyk, Divya Situt, Sheryl Beauchamp, Jawaria Abdali, Anu Dinesh, Nathalie Legancher, Hongyu Sun, Michael Schlossmacher, Shawn Hayley; CLINT (Canadian LRRK2 in inflammation team) (2020). Microglia depletion prior to lipopolysaccharide and paraquat treatment differentially modulates behavioral and neuronal outcomes in wild type and G2019S LRRK2 knock-in mice. Brain Behavior and Immunity-Health, Volume 5, May, 100079.
- Zach Dwyer ,Chris Rudyk, Ashley Thompson, Kyle Farmer, Barbara Fenner, Teresa Fortina, Alexa Derksen, Hongyu Sun, Shawn Hayley; CLINT (Canadian LRRK2 in inflammation team) (2020). Leucine rich repeat kinase-2 (LRRK2) modulates microglial phenotype and dopaminergic neurodegeneration. Neurobiology of Aging, 91:45-55.
- Kyle Farmer, Alexa Derksen, Elyn M. Rowe, Ashley M. Thompson, Christopher A. Rudyk, Natalie A. Prowse, Zachary Dwyer, Teresa Fortin, Khaled S. Abd-Elrahman, Stephen S.G. Ferguson, Shawn Hayley (2020). mGluR5 modulation promotes neurorecovery through mTOR in a Parkinson's model. Mol Neurobiology, 57(3):1418-1431.
- Natalie Prowse, Zach Dwyer, Amanda Thompson, Teresa Fortin, Kristin Elson, H. Robeson, Barbara Fenner, Shawn Hayley (2020). Early life selective knockdown of the TrkB receptor modulates adult stress phenotype. Behavioral Brian Research, 27;378:112260.
- Barbara Fenner, Natalie Prowse, Teresa Fortin, Shawn Hayley (2020). A novel dual-hit in vitro model of Parkinson’s disease: WAVE2-dependent progression of microglial inflammatory phenotype. Journal of Cell Biology, Submitted.
- Zach Dwyer, Chris Rudyk, Divya Situt, Sheryl Beauchamp, Jawaria Abdali, Anu Dinesh, Nathalie Legancher, Hongyu Sun, Michael Schlossmacher, Shawn Hayley; CLINT (Canadian LRRK2 in inflammation team) (2020). Microglia depletion prior to lipopolysaccharide and paraquat treatment differentially modulates behavioral and neuronal outcomes in wild type and G2019S LRRK2 knock-in mice. Journal of Neuroinflammation, in revision.
- Zach Dwyer, Chris Rudyk, Kyle Farmer, Sheryl Beauchamp, Pragya Shail, Alexa Derksen, Teresa Fortin, Katelyn Ventura, Carlos Torresd, Kiara Ayoub Shawn Hayley PhD (2020). Characterizing the protracted neurobiological and neuroanatomical effects of paraquat in a murine model of Parkinson’s disease. Neurobiology of Aging, in revision.
- Zach Dwyer ,Chris Rudyk, Ashley Thompson, Kyle Farmer, Barbara Fenner, Teresa Fortina, Alexa Derksen, Hongyu Sun, Shawn Hayley; CLINT (Canadian LRRK2 in inflammation team) (2019). Leucine rich repeat kinase-2 (LRRK2) modulates microglial phenotype and dopaminergic neurodegeneration. Neurobiology of Aging, in press.
- Kyle Farmer, Alexa Derksen, Elyn M. Rowe, Ashley M. Thompson, Christopher A. Rudyk, Natalie A. Prowse, Zachary Dwyer, Teresa Fortin, Khaled S. Abd-Elrahman, Stephen S.G. Ferguson, Shawn Hayley (2019). mGluR5 modulation promotes neurorecovery through mTOR in a Parkinson's model. Mol neurobiology, doi: 10.1007/s12035-019-01818-z.
- Natalie Prowse, Zach Dwyer, Amanda Thompson, Teresa Fortin, Kristin Elson, H. Robeson, Barbara Fenner, Shawn Hayley (2019). Early life selective knockdown of the TrkB receptor modulates adult stress phenotype. Behavioral Brian Research, 27;378:112260.
- Jeffery Landrigan, Zach Dwyer, Sheryl Beauchamp, Shawn Hayley (2019). Quantum dot conjugated saporin activates microglia and induces selective substantia nigra degeneration. Neurotoxicology, 76:153-161.
- Chris Rudyk, Zach Dwyer, McNeill J, Salmaso N, Kyle Farmer, Prowse N, Shawn Hayley (2019). Chronic unpredictable stress influenced the behavioral but not the neurodegenerative impact of paraquat. Neurobiology of Stress, 11:100179.
- Chris Rudyk, Zach Dwyer, Ashley Thompson, Kyle Farmer, Teresa Fortina, Alexa Derksen, Shawn Hayley; CLINT (Canadian LRRK2 in inflammation team) (2019). Leucine rich repeat kinase-2 (LRRK2) modulates paraquat induced inflammatory sickness and stress phenotype. Journal of Neuroinflammation, 16(1):120.
- Bojan Shutinoski †1,2, Mansoureh Hakimi †1,3, Irene E. Harmsen 1, Michaela Lunn 1,4, Juliana Rocha 8, Nathalie Lengacher 1, Yi Yuan Zhou 1, Jasmine Khan 1, Angela Nguyen 1, Quinton Hake-Volling 1, Daniel El-Kodsi 1, Juan Li 1, Azadeh Alikashani 9,10, Claudine Beauchamp 9,10, Jay Majithia 2, Kevin Coombs 11, Paul C. Marcogliese 3,6, David S. Park 3,6, John D. Rioux 9,10, Dana Philpott 8, John M. Woulfe 1,5, Shawn Hayley 4, Subash Sad 2, Julianna J. Tomlinson 1,6, Earl G. Brown 1,2, Canadian LRRK2 in Inflammation Team (CLINT)12 and Michael G. Schlossmacher (2019). Lrrk2 Alleles Modulate Inflammation during Infections in a Pathogen-, Organ- and Sex-Dependent Manner. Science Translational Medicine; 11(511).
- Jadavji NM, Murray LK, Emmerson JT, Rudyk CA, Hayley S, Smith PD. (2019). Paraquat exposure increases oxidative stress within the dorsal striatum of male mice with a genetic deficiency in one-carbon metabolism. Toxicol Sci. 169(1):25-33.
- Boutin, R Dwyer Z, Farmer K, Rudyk C, Forbes M*, Hayley S* (2018) PERINATAL ANTIBIOTIC EXPOSURE ALTERS COMPOSITION OF MURINE GUT MICROBIOTA AND MAY INFLUENCE LATER RESPONSES TO PEANUT ANTIGEN. Allergy, Asthma & Clinical Immunology, in press.
- Rudyk* C, Dwyer* Z, Thompson A, Farmer K, Fortin T, Derksen A, Hayley S. (2018) Leucine rich repeat kinase-2 (LRRK2) modulates microglial phenotype and dopaminergic neurodegeneration. NPJ Parkinson, In revision.
- Litteljohn D, Rudyk C, Dwyer Z, Farmer K, Fortin T, Hayley S; Canadian LRRK2 in Inflammation Team (CLINT) (2018). The impact of murine LRRK2 G2019S transgene overexpression on acute responses to inflammatory challenge. Brain Behav Immun. Jan;67:246-256
- Rudyk CA, McNeill J, Prowse N, Dwyer Z, Farmer K, Litteljohn D, Caldwell W, Hayley S. (2017). Age and Chronicity of Administration Dramatically Influenced the Impact of Low Dose Paraquat Exposure on Behavior and Hypothalamic-Pituitary-Adrenal Activity. Front Aging Neurosci. Jul 14;9:222.
- Litteljohn D, Rudyk C, Razmjou S, Dwyer Z, Syed S, Hayley S. (2017). Individual and interactive sex-specific effects of acute restraint and systemic IFN-γ treatment on neurochemistry. Neurochem Int.;102:95-104.
- Lau C, Hebert M, Vani MA, Walling S, Hayley S, Lagace DC, Blundell J. (2016). Absence of neurogenic response following robust predator-induced stress response. Neuroscience.;339:276-286.
- Clarke M, Razmjou S, Prowse N, Dwyer Z, Litteljohn D, Pentz R, Anisman H, Hayley S. (2017). Ketamine modulates hippocampal neurogenesis and pro-inflammatory cytokines but not stressor induced neurochemical changes. Neuropharmacology. 2016 Apr 20. pii: S0028-3908(16)30161-7.
- Hayley S and Anisman (2016). Inflammation and the microbiome: Implications for depressive disorders. Curr Opin Pharm, 29:42-6.
- Ramzjou S, Litteljohn D, Dwyer Z, Hayley S (2016). The interactive effects of ketamine and magnesium upon depressive-like pathology. Neuro Psych Dis, 12:2049-56.
- Landrigan J, Shwarf F, Abaziad A, Hayley S (2016). Interactive effects of ghrelin and ketamine on forced swim performance: Implications for novel antidepressant strategies. Neurosci Lett, submitted.
- Litteljohn D, Ramzjou S, Dwyer Z, Hayley S (2016). LRRK2 G2019S transgenic overexpression had little impact on the acute central effects of LPS or IFN-γ treatment. Eur J Neurosci, submitted.
- Rudyk C, Litteljohn D, Syed S, Dwyer Z, Hayley S (2015). Paraquat and psychological stressor interactions as pertains to Parkinsonian co-morbidity. Neurobiol Stress, 2:85-93.
- Rudyk C, Litteljohn D, Liu G, Hayley (2015). Unexpected hormetic-like effects of long-term low-dose paraquat exposure in aged mice. Neuro Tox, in revision.
- Farmer K, Smith CA, Hayley S, Smith J. (2015). Major Alterations of Phosphatidylcholine and Lysophosphotidylcholine Lipids in the Substantia Nigra Using an Early Stage Model of Parkinson's Disease. Int J Mol Sci. 2015 Aug 12;16(8):18865-77.
- Hayley S, Du L, Poulter MO, Anisman H (2015). Gender and brain regions specific differences in brain derived neurotrophic factor protein levels of depressed individuals that died through suicide. Neurosci Lett, 600:12-6.
- Farmer K, Rudyk C, Prowse NA, Hayley S (2015). Granulocyte macrophage-colony stimulating factor and erythropoietin influence striatal regeneration in a prodromal model of Parkinson’s disease. Front Cell Neurosci, 7:126. doi: 10.3389/fnagi.2015.00126.
- Litteljohn D, Nelson E, Hayley S (2014). IFN-g differentially modulates memory-related processes under basal and chronic stressor conditions. Front cell Neurosci, 8:391. doi: 10.3389/fncel.2014.00391.
- Hayley S (2014). The neuroiummune-neuroplasticity interface and brain pathology. Front Cell Neurosci, 4;8:419. doi: 10.3389/fncel.2014.00419.
- Serafini G, Hayley S, Dwivedi Y, Girardi P, Amore M (2014). Hippocampal neurogenesis, neurotrophic factors and depression: possible therapeutic targets. CNS Neurol Disor Drug Targets, 13(10):1708-21.
- Liu G, Rustom N, Litteljohn D, Rudyk C, Anisman H, Hayley S (2014). Use of induced pluripotent stem cell derived neurons engineered to express BDNF for modulation of stressor related pathology. Front Cell Neurosci; Oct 14; doi: 10.3389/fncel.2014.00316.
- Liu G1, Anisman H, Bobyn J, Hayley S. (2014) Interaction Between Nonviral Reprogrammed Fibroblast Stem Cells and Trophic Factors for Brain Repair. Mol Neurobiol. 50(2):673-84.
- Hayley S, Anisman H. (2013). Neurotrophic paths in the treatment of depression. J Psychiatry Neurosci. 38(5):291-3. doi: 10.1503/jpn.130146.
- Osborn M, Rustom N, Clarke M, Litteljohn D, Rudyk C, Anisman H, Hayley S* (2013). Antidepressant-like effects of erythropoietin: A focus on hippocampal and behavioral processes. Plos One, 8(9):e72813.
- *Caldwell W, McInnis O, McQuaid R, Liu G, Stead J, Anisman H, Hayley S (2013). The Role of the Val66Met Polymorphism of the Brain Derived Neurotrophic Factor Gene in Coping Strategies Relevant to Depressive Symptoms. Plos One, Sep 3;8(9):e72813. *This paper was used in a meta-analyses on depression and neurotrophic factors: “Hosang, GM, Shiles, C, Tansay, KE, McGuffin, P & Uher, R. (2014). Interaction between stress and the BDNF Val66Met polymorphism in depression: a systematic review and meta-analysis. BMC Medicine 12: DOI: 10.1186/1741-7015-12-7
- Hayley S and Litteljohn D (2013). Neuroplasticity and the next wave of anti-depressant strategies. Front Cell Neurosci, 7:218. doi: 10.3389/fncel.2013.00218.
- Sliz D, Hayley S. (2013). Major depressive disorder and alterations in insular cortical activity: a review of current functional magnetic imaging research. Front Hum Neurosci.6:323.
- Anisman H, Hayley S. (2012). Inflammatory factors contribute to depression and its comorbid conditions. Sci Signal. Oct 2;5(244):pe45. doi: 10.1126/scisignal.2003579.
- Clarke M, Pentz R, Bobyn J, Hayley S. (2012). Stressor-like effects of c-Jun N-terminal kinase (JNK) inhibition. PLoS One.;7(8):e44073. doi: 10.1371/journal.pone.0044073.
- Litteljohn D, Hayley S. (2012) Cytokines as potential biomarkers for Parkinson's disease: a multiplex approach. Methods Mol Biol.;934:121-44. doi: 10.1007/978-1-62703-071-7_7.
- Litteljohn D, Hayley S (2012). Inflammatory roads to neurodegeneration: A focus on Parkinson’s and Alzheimer’s disease. Hbk Psychoneuro;Jul 8, 393-410.
- Hayley S, Scharf J, Anisman H. (2012) Central administration of murine interferon-α induces depressive-like behavioral, brain cytokine and neurochemical alterations in mice: A mini-review and original experiments. Brain Behav Immun. 31:115-27.
- Anisman H, Hayley S. (2012) Illness comorbidity as a biomarker? J Psychiatry Neurosci. 37(4):221-3. doi: 10.1503/jpn.120092.
- Bobyn J, Mangano EN, Gandhi A, Nelson E, Moloney K, Clarke M, Hayley S. (2012). Viral-toxin interactions and Parkinson's disease: poly(I:C) priming enhanced the neurodegenerative effects of paraquat. J Neuroinflammation. 4;9:86. doi: 10.1186/1742-2094-9-86
- Sliz D, Smith A, Wiebking C, Northoff G, Hayley S (2012). Neural correlates of a single-session massage treatment. Brain Imaging Beh. 6(1):77-87.
- Gibb J, Hayley S, Poulter MO, Anisman H. (2011). Effects of stressors and immune activating agents on peripheral and central cytokines in mouse strains that differ in stressor responsivity. Brain Behav Immun; 25(3):468-82.
- Mangano EN, Peters S, Litteljohn D, So R, Bethune C, Bobyn J, Clarke M, Hayley S. (2011). Granulocyte macrophage-colony stimulating factor protects against substantia nigra dopaminergic cell loss in an environmental toxin model of Parkinson's disease. Neurobiol Dis. 43(1):99-112.
- Mangano EN, Litteljohn D, So R, Nelson E, Peters S, Bethune C, Bobyn J, Hayley S. (2011). Interferon-γ plays a role in paraquat-induced neurodegeneration involving oxidative and proinflammatory pathways. Neurobiol Aging. 33(7):1411-26.
- Hayley S. (2011). Toward an anti-inflammatory strategy for depression. Front Behav Neurosci. 13;5:19.
- Hayley S, Mangano E, Crowe G, Li N, Bowers WJ. (2011). An in vivo animal study assessing long-term changes in hypothalamic cytokines following perinatal exposure to a chemical mixture based on Arctic maternal body burden. Environ Health. 11;10:65.
- Litteljohn D, Nelson E, Bethune C, Hayley S. (2011). The effects of paraquat on regional brain neurotransmitter activity, hippocampal BDNF and behavioural function in female mice. Neurosci Lett. 502(3):186-91.
- Litteljohn D, Mangano E, Clarke M, Bobyn J, Moloney K, Hayley S. (2010). Inflammatory mechanisms of neurodegeneration in toxin-based models of Parkinson's disease. Parkinsons Dis., 2011:713517.
- Hayley S (2010). A comment on the role of environmental factors in Parkinson's disease. Can J Neurol Sci. 37(5):555-6
- Litteljohn D, Cummings A, Brennan A, Gill A, Chunduri S, Anisman H, Hayley S. (2010). Interferon-gamma deficiency modifies the effects of a chronic stressor in mice: Implications for psychological pathology. Brain Behav Immun.;24(3):462-73.
- Anisman H, Hayley S (2010). Cytokine effects on neuronal processes and on behavior. Enc Beh Neurosi, 1, 361-369.
- Merali Z, Hayley S, Kent P, McIntosh J, Bédard T, Anisman H. (2009). Impact of repeated stressor exposure on the release of corticotropin-releasing hormone, arginine-vasopressin and bombesin-like peptides at the anterior pituitary. Behav Brain Res. 198(1):105-12.
- Seguin JA, Mangano EN, Brennan J & Hayley S (2009). Proinflammatory cytokines differentially influence adult hippocampal cell proliferation depending upon the route and chronicity of administration. Neuropsy. Dis. Treatment, 5, 5-14.
- Litteljohn D, Mangano E, Shukla N, Hayley S. (2009). Interferon-gamma deficiency modifies the motor and co-morbid behavioral pathology and neurochemical changes provoked by the pesticide paraquat. Neuroscience. 164(4):1894-906.
- Gibb J, Audet MC, Hayley S, Anisman H. (2009). Neurochemical and behavioral responses to inflammatory immune stressors. Front Biosci (Schol Ed). 1;1:275-95.
- Litteljohn D, Mangano EN & Hayley S (2008). Cyclooxygenase-2 deficiency modifies the neurochemical effects, motor impairment and co-morbid anxiety provoked by paraquat administration in mice. Eur. J. Neurosci., 28(4):707-16.
- Kentner AC, Takeuchi A, James JS, Miki T, Seino S, Hayley S, Bielajew C. (2008). The effects of rewarding ventral tegmental area stimulation and environmental enrichment on lipopolysaccharide-induced sickness behavior and cytokine expression in female rats. Brain Res. 1217:50-61.
- Anisman H, Merali Z & Hayley S (2008). Neurotransmitter, peptide and cytokine processes in relation to depressive disorder: Comorbidity between depression and neurodegenerative disorders. Progress in Neurobiol, 85(1):1-74.
- Mangano E.N., Hayley S. (2009). Inflammatory priming of the substantia nigra influences the impact of later paraquat exposure: Neuroimmune sensitization of neurodegeneration. Neurobiology of Aging, 30(9):1361-78.
- Anisman H, Gibb J, Hayley S. (2008). Influence of continuous infusion of interleukin-1beta on depression-related processes in mice: corticosterone, circulating cytokines, brain monoamines, and cytokine mRNA expression. Psychopharmacology 199(2):231-44.
- Hayley S, Mangano EN, Strickland M & Anisman H (2008). Lipopolysaccharide and a social stressor influence behaviour, corticosterone and cytokine levels: Divergent actions in cyclooxygenase-2 deficient mice and wild type controls. J. Neuroimmunol., 197(1):29-36.
- Gibb J, Hayley S, Gandhi R, Poulter MO, Anisman H. (2008). Synergistic and additive actions of a psychosocial stressor and endotoxin challenge: Circulating and brain cytokines, plasma corticosterone and behavioral changes in mice. Brain Beh Immun, 22(4):573-89.
- Mount M, Lira A, Alyeason H, Grimes D, Smith P, Slack R, Anisman H, Hayley S*, Park DS* (2007). Central nature of interferon-gamma in microglial mediated loss of dopaminergic neurons. J. Neurosci, 27, 3328-3337. *Co-senior authors contributed equally to this manuscript.
- Anisman H, Poulter MO, Gandhi R, Merali Z, Hayley S (2007). Interferon-effects are exaggerated when administered on a psychosocial stressor backdrop: Cytokine, corticosterone and brain monoamine variations. J. Neuroimmunol., 186(1-2):45-53.
- Gandhi R, Hayley S, Gibb J, Merali Z, Anisman H. (2007). Influence of poly I:C on sickness behaviors, plasma cytokines, corticosterone and central monoamine activity: Moderation by social stressors. Brain Behav Immun. 21(4):477-89.
- Crocker S.J*. Hayley S*., Smith P.D., Callaghan S.M., Slack R.S., & Park D.S. (2006). Stress-Activated MAP Kinase Signaling in Nigral Dopamine Neurons Modifies Axotomy-Induced c-Jun Expression and Neurodegeneration in vivo. J. Neurochem., 96, 489-499. *authors contributed equally.
- Hayley S, Poulter MO, Merali Z & Anisman H (2005). The pathogenesis of clinical depression: stressor- and cytokine-induced alterations of neuroplasticity. Neuroscience. 135, 659-678.
- Kim RH, Smith PD, Aleyasin H, Hayley S, Mount MP, Pownall S, Wakeham A, You-Ten AJ, Kalia SK, Horne P, Westaway D, Lozano AM, Anisman H, Park DS, Mak TW.(2005). Hypersensitivity of DJ-1-deficient mice to 1-methyl-4-phenyl-1,2,3,6-tetrahydropyrindine (MPTP) and oxidative stress. Proc. Natl. Acad. Sci. 102(14):5215-20.
- Hayley S, Anisman H. (2005). Multiple mechanisms of cytokine action in neurodegenerative and psychiatric states: neurochemical and molecular substrates. Curr Pharm Des. 11(8):947-62.
- Anisman H, Merali Z, Poulter MO, Hayley S (2005). Cytokines as a precipitant of depressive illness: animal and human studies. Curr Pharm Des. 11(8):963-72.
- Sudom K, Turrin NP, Hayley S, Anisman H. (2004). Influence of chronic interleukin-2 infusion and stressors on sickness behaviors and neurochemical change in mice. Neuroimmunomodulation. 11(5):341-50.
- Hayley S, Kelly OP & Anisman (2004). Corticosterone changes in response to stressors, acute and protracted actions of tumor necrosis factor-, and lipopolysaccharide treatments in mice lacking the tumor necrosis factor- p55 receptor gene. Neuroimmunomodulation. 11(4):241-6.
- Hayley S, Crocker SJ, Smith P, Shree T & Park DS (2004). Regulation of dopaminergic loss by Fas in a 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine model of Parkinson's disease. J. Neurosci., 24, 2045-2053.
- Anisman H, Turrin N, Merali Z & Hayley S (2003). Neurochemical Sensitization Associated with Systemic administration of Tumor Necrosis Factor-: Adjuvant Action in Combination with Bovine Serum Albumin. J. Neuroimmunol., 145, 91-102.
- Smith, P, Crocker SJ, Jackson-Lewis V, Prezborski S, Hayley S, Slack R, & Park DS (2003). Neuroprotective effects of CDK5 in the MPTP model of Parkinson’s disease. Proc. Natl. Acad. Sci., 100, 13650-13655.
- Crocker SJ, Smith P, Jackson-Lewis V, Hayley S, Prezborski S, Lambda W, Slack R & Park DS (2003). Role of calpain in MPTP-induced dopamine neuron loss and behavioural impairment in mice. J. Neurosci., 23, 4081-4091.
- Anisman H, Merali Z & Hayley S (2003). Sensitization associated with stressors and cytokine treatments. Brain Behav. Immun., 17, 86-93.
- Michaud DS McLean J, Keith SE Ferrarotto C, Hayley S, Anisman H & Merali Z (2003). Strain differences in neurochemical and neuroendocrine responses to an acute audiogenic stressor in male Fischer 344 and Lewis rats. Neuropsychopharmacol., 28, 1068-1081.
- Hayley S, Merali Z & Anisman H (2003). Stress and cytokine-elicited neuroendocrine and neurotransmitter sensitization: implications for depressive illness. Stress, 6, 19-32.
- Anisman H, Hayley S, Turrin N, Merali Z. Cytokines as a stressor: implications for depressive illness. Int J Neuropsychopharmacol. 2002 Dec;5(4):357-73.
- Hayley S, Kelly O, Anisman H. Murine tumor necrosis factor-alpha sensitizes plasma corticosterone activity and the manifestation of shock: modulation by histamine. J Neuroimmunol. 2002 Oct;131(1-2):60-9.
- Hayley S, Wall P, Anisman H. (2002). Sensitization to the neuroendocrine, central monoamine and behavioural effects of murine tumor necrosis factor-alpha: peripheral and central mechanisms. Eur J Neurosci. 15(6):1061-76.
- Hayley S, Staines W, Merali Z, Anisman H. (2001). Time-dependent sensitization of corticotropin-releasing hormone, arginine vasopressin and c-fos immunoreactivity within the mouse brain in response to tumor necrosis factor-alpha. Neuroscience.;106(1):137-48.
- Anisman H, Hayley S, Kelly O, Borowski T, Merali Z. (2001). Psychogenic, neurogenic, and systemic stressor effects on plasma corticosterone and behavior: mouse strain-dependent outcomes. Behav Neurosci. 115(2):443-54.
- Hayley S, Lacosta S, Merali Z, van Rooijen N, Anisman H. (2001). Central monoamine and plasma corticosterone changes induced by a bacterial endotoxin: sensitization and cross-sensitization effects. Eur J Neurosci. 13(6):1155-65.
- Hayley S, Borowski T, Merali Z, Anisman H. (2001); Central monoamine activity in genetically distinct strains of mice following a psychogenic stressor: effects of predator exposure. Brain Res. 892(2):293-300.
- Anisman H, Kelly O, Hayley S, Borowski T, Merali Z, McIntyre DC. (2000). Acoustic startle and fear-potentiated startle in rats selectively bred for fast and slow kindling rates: relation to monoamine activity. Eur J Neurosci. 12(12):4405-16.
- Brebner K, Hayley S, Zacharko R, Merali Z, Anisman H. (2000). Synergistic effects of interleukin-1beta, interleukin-6, and tumor necrosis factor-alpha: central monoamine, corticosterone, and behavioral variations. Neuropsychopharmacology. 22(6):566-80.
- McIntyre DC, Kent P, Hayley S, Merali Z, Anisman H. (1999). Influence of psychogenic and neurogenic stressors on neuroendocrine and central monoamine activity in fast and slow kindling rats. Brain Res. 840(1-2):65-74.
- Hayley S, Brebner K, Lacosta S, Merali Z, Anisman H. (1999). Sensitization to the effects of tumor necrosis factor-alpha: neuroendocrine, central monoamine, and behavioral variations. J Neurosci. 19(13):5654-65.